Perspective | Catheter-Directed Management in Acute Pulmonary Embolism
Pulmonary embolism (PE) is a common cause of cardiovascular death, with an increasing incidence.1-4 The insult of an acute increase in right ventricular (RV) afterload secondary to thrombus obstruction in the pulmonary arteries, hypoxemic vasoconstriction and release of pulmonary artery vasoconstrictors can lead to RV dysfunction and dilation and decreased left ventricular (LV) preload, with resultant cardiovascular collapse in the most severe cases.5 Anticoagulation is the mainstay of treatment of all PEs with excellent outcomes in low-risk patients.6,7
There is a subset of patients with intermediate-high and high-risk PE, however, that may benefit from more advanced therapy, including systemic fibrinolysis, catheter-directed therapy (CDT) with or without infusion of fibrinolytics or pulmonary embolectomy, as these options actively reduce thrombus burden to decrease RV afterload. In those patients who survive the initial presentation, long-term sequelae can develop including persistent RV dysfunction, chronic thromboembolic pulmonary hypertension, and decreased quality of life and exercise tolerance.8,9
Systemic fibrinolysis has been proven to quickly decrease thrombus burden and pulmonary artery pressures. However, this is at the risk of a major bleeding rate that exceeds 20 percent and the feared complication of intracranial hemorrhage (ICH) in 3 percent.10,11 Concern for bleeding complications has led to the underutilization of systemic fibrinolysis, with a high proportion of patients possessing an absolute or relative contraindication.12 Given the rate of adverse events, systemic fibrinolysis has generally been limited to those at highest risk with evidence of hemodynamic compromise or cardiogenic shock. CDT has been increasingly utilized to offer active improvement in RV afterload beyond anticoagulation alone, but with a reduced dose or no fibrinolytic therapy.
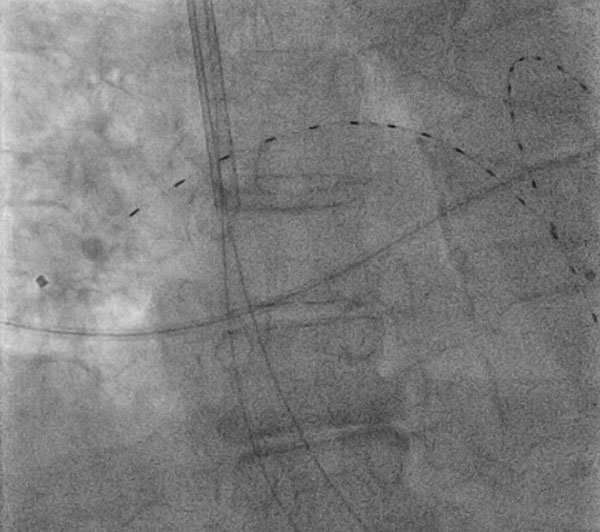 Figure 1: Catheter-Directed, Ultrasound-Facilitated, Low-Dose Fibrinolysis
Figure 1: Catheter-Directed, Ultrasound-Facilitated, Low-Dose FibrinolysisA 74-year-old woman presented with bilateral proximal pulmonary embolism with persistent tachycardia and received catheter-directed, ultrasound-facilitated, low-dose fibrinolysis under fluoroscopic guidance via right internal jugular vein access. The device pictured is the EkoSonic Endovascular System (EKOS, Bothell, WA) in the bilateral pulmonary arteries.
CDTs include those that deliver fibrinolytic agents (i.e., tissue plasminogen activator [t-PA]) with or without ultrasound assistance, as well as devices that decrease clot burden by a variety of mechanical methods without fibrinolytic agents. The best studied and only U.S. Food and Drug Administration-approved catheter-directed device for management of PE is the EkoSonic Endovascular System (EKOS, Bothell, WA) (Figure 1). The 5.4 French infusion catheters are placed directly into the pulmonary arteries, usually via internal jugular or femoral vein access, and contain an ultrasonic core that releases ultrasound waves in addition to a series of pores that deliver t-PA.
In vitro studies have demonstrated improved penetration and binding of t-PA to fibrin in the presence of ultrasound.13 In a small trial of 59 patients with intermediate-risk PE randomized to ultrasound-facilitated, low-dose fibrinolysis plus anticoagulation vs. anticoagulation alone, the intervention group had a greater decrease in RV-to-LV diameter ratio and pulmonary artery systolic pressures, and a more significant improvement in RV function at 24 hours.14 At 90-day follow-up, there was a trend toward greater reduction in RV size in the intervention group (p = 0.07); however, there was no difference in systolic artery pressure.
"CDT has been increasingly utilized to offer active improvement in RV afterload beyond anticoagulation alone, but with a reduced dose or no fibrinolytic therapy."
A larger, single-arm, multicenter trial evaluated ultrasound-facilitated, low-dose fibrinolysis in 150 patients with massive or submassive PE. There was a significant reduction in RV-to-LV diameter ratio, pulmonary artery systolic pressure and thrombus burden at 48 hours.15 There was one severe and 15 moderate bleeding events, with no intracranial hemorrhage. Infusion catheters without ultrasound (e.g., Cragg-McNamara, pigtail catheter) also offer delivery of fibrinolytic into the pulmonary arteries with significantly less cost compared with ultrasound-facilitated fibrinolysis. Though limited to small retrospective studies, catheter-directed fibrinolysis with and without ultrasound have been evaluated and show similar outcomes.16,17 These findings have not been validated in a randomized-controlled trial, though a trial is ongoing (NCT02758574).18
A meta-analysis evaluated 16 studies of catheter-directed thrombolysis and found a major complication rate of 4.7 percent and intracranial hemorrhage rate of 0.35 percent.19 A recent propensity-matched analysis of in-hospital outcomes utilized the National Readmission Database to compare systemic vs. catheter-directed fibrinolysis and demonstrated lower in-hospital mortality and bleeding in patients who received catheter-directed fibrinolysis.20 There have been no prospective, randomized trials comparing systemic to catheter-directed fibrinolysis.
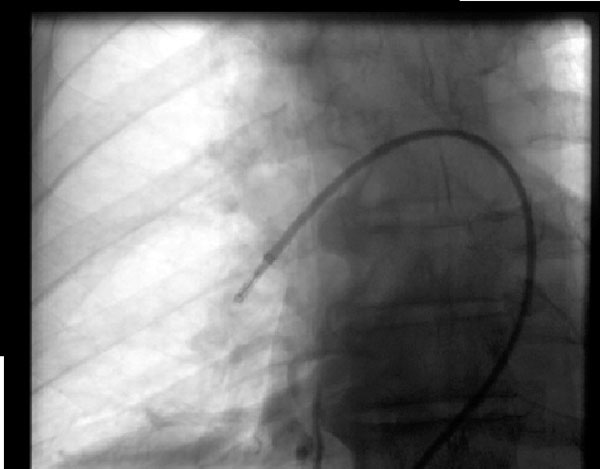 Figure 2: Aspiration Thrombectomy
Figure 2: Aspiration ThrombectomyA 68-year-old man developed a massive pulmonary embolism one day after spine surgery and was not a candidate for fibrinolysis, thus underwent aspiration thrombectomy under fluoroscopic guidance with improvement in his hemodynamics. The device pictured is the Indigo embolectomy system (Penumbra, Alameda, CA) in the right pulmonary artery.
Frequently, patients will warrant advanced therapy but have contraindications to even low doses of t-PA. In such patients, pulmonary embolectomy is a possible option. However, it requires a sternotomy and cardiopulmonary bypass with high-doses of heparin. Mechanical CDT offers a percutaneous option that does not utilize fibrinolysis. There are various mechanical CDTs including aspiration or suction mechanical thrombectomy (Figure 2), maceration with a pigtail catheter, and rheolytic thrombectomy.
There are various limitations to each available system such as adequate manipulation into the pulmonary arteries due to catheter size, blood loss during aspiration and limited ability to remove thrombus, particularly if it is older and more fibrotic. There may be a role for combined mechanical and fibrinolytic therapy for improved thrombus clearance.21 Data for mechanical CDT is limited to small, single-center case series, though demonstrate reasonable technical success.22-25 CDT also offers a bailout treatment option for patients with persistent shock despite systemic fibrinolysis or patients who have been placed on extracorporeal membrane oxygenation.26
"There are no high-quality studies comparing the various CDT against each other or against systemic thrombolysis."
There are no high-quality studies comparing the various CDT against each other or against systemic thrombolysis. Given the current rate of CDT use in intermediate-risk PE and the potential risks and costs of these interventions, evaluation of long-term outcomes, including pulmonary hypertension and exercise tolerance, in addition to higher quality short-term clinical outcome data are necessary.
In conclusion, CDT is a promising management option for patients with acute PE and offers an alternative advanced therapy for patients who are poor candidates for systemic thrombolysis or pulmonary embolectomy. It remains an exciting field with rapid development of new therapies, but requires further research prior to its widespread utilization in intermediate-risk PE.
Brett Carroll, MD, recently completed his Vascular Medicine Fellowship at Brigham and Women’s Hospital and is now the director of Vascular Medicine at Beth Israel Deaconess Medical Center, both in Boston, MA.
References
- Jimenez D, de Miguel-Diez J, Guijarro R, et al. J Am Coll Cardiol 2016;67:162-70.
- Huang W, Goldberg RJ, Anderson FA, et al. Am J Med 2014;127:829-39 e5.
- Cohen AT, Agnelli G, Anderson FA, et al. Thromb Haemost 2007;98:756-764.
- Mozaffarian D, Benjamin EJ, Go AS, et al. Circulation 2016;133:e38-360.
- Piazza G, Goldhaber SZ. Circulation 2010;122:1124-9.
- Aujesky D, Obrosky S, Stone RA, et al. Am J Resp Crit Care Med 2005;172:1041-6.
- Kearon C, Akl EA, Ornelas J, et al. Chest 2016;149:315-52.
- Piazza G, Goldhaber SZ. N Eng J Med 2011;364:351-60.
- Sista AK, Miller LE, Kahn SR, Kline JA. Vasc Med 2017;22:37-43.
- Goldhaber SZ, Haire WD, Feldstein ML, et al. Lancet 1993;341:507-11.
- Goldhaber SZ, Visani L, De Rosa M. Lancet 1999;353:1386-9.
- Stein PD, Matta F. Am J Med 2012;125:465-70.
- Siddiqi F, Odrljin TM, Fay PJ, et al. Blood 1998;91:2019-25.
- Kucher N, Boekstegers P, Muller OJ, et al. Circulation 2014;129:479-86.
- Piazza G, Hohlfelder B, Jaff MR, et al. JACC Cardiovasc Interv 2015;8:1382-92.
- Liang NL, Avgerinos ED, Marone LK, et al. Vasc Endovascular Surg 2016;50:405-10.
- Graif A, Grilli CJ, Kimbiris G, et al. J Vasc Interv Radiol 2017;28:1339-47.
- Avgerinos ED, Mohapatra A, Rivera-Lebron B, et al. J Vasc Surg Venous Lymphat Disord 2018;6:126-32.
- Bloomer TL, El-Hayek GE, McDaniel MC, et al. Catheter Cardiovasc Interv 2017;89:754-60.
- Arora S, Panaich SS, Ainani N, et al. Am J Cardiol 2017;120:1653-61.
- Liu B, Liu M, Yan L, et al. J Int Med Res 2017;Jan 1; [Epub ahead of print].
- Ciampi-Dopazo JJ, Romeu-Prieto JM, Sanchez-Casado M, et al. J Vasc Interv Radiol 2018;29:101-6.
- Tukaye DN, McDaniel M, Liberman H, et al. JACC Cardiovasc Interv 2017;10:94-5.
- Hubbard J, Saad WE, Sabri SS, et al. Thrombosis 2011;2011:246410.
- Donaldson CW, Baker JN, Narayan RL, et al. Catheter Cardiovasc Interv 2015;86:e81-7.
- George B, Parazino M, Omar HR, et al. Resuscitation 2018;122:1-5.
Clinical Topics: Anticoagulation Management, Arrhythmias and Clinical EP, Cardiac Surgery, Diabetes and Cardiometabolic Disease, Dyslipidemia, Heart Failure and Cardiomyopathies, Invasive Cardiovascular Angiography and Intervention, Prevention, Pulmonary Hypertension and Venous Thromboembolism, Vascular Medicine, SCD/Ventricular Arrhythmias, Atrial Fibrillation/Supraventricular Arrhythmias, Cardiac Surgery and Arrhythmias, Cardiac Surgery and Heart Failure, Lipid Metabolism, Statins, Acute Heart Failure, Pulmonary Hypertension, Interventions and Vascular Medicine, Hypertension
Keywords: ACC Publications, Cardiology Interventions, United States Food and Drug Administration, Blood Pressure, Cardiopulmonary Bypass, Dilatation, Embolectomy, Exercise Tolerance, Extracorporeal Membrane Oxygenation, Femoral Vein, Fibrin, Fibrinolysis, Fibrinolytic Agents, Follow-Up Studies, Heparin, Hospital Mortality, Hypertension, Pulmonary, Indigo Carmine, Intracranial Hemorrhages, Jugular Veins, Patient Readmission, Pulmonary Artery, Pulmonary Embolism, Quality of Life, Retrospective Studies, Shock, Cardiogenic, Sternotomy, Suction, Tachycardia, Thrombectomy, Thrombolytic Therapy, Thrombosis, Tissue Plasminogen Activator, High-Energy Shock Waves, Vasoconstriction, Vasoconstrictor Agents
< Back to Listings


